CM Presentation Date: November 18, 2021 @ 2:00 PM in Pathology 103
Title: “Understanding the Contribution of HIV Autoprocessing to PI Resistance”
Advisor: Chaoping Chen

CM Presentation Date: November 18, 2021 @ 2:00 PM in Pathology 103
Title: “Understanding the Contribution of HIV Autoprocessing to PI Resistance”
Advisor: Chaoping Chen
ABSTRACT:
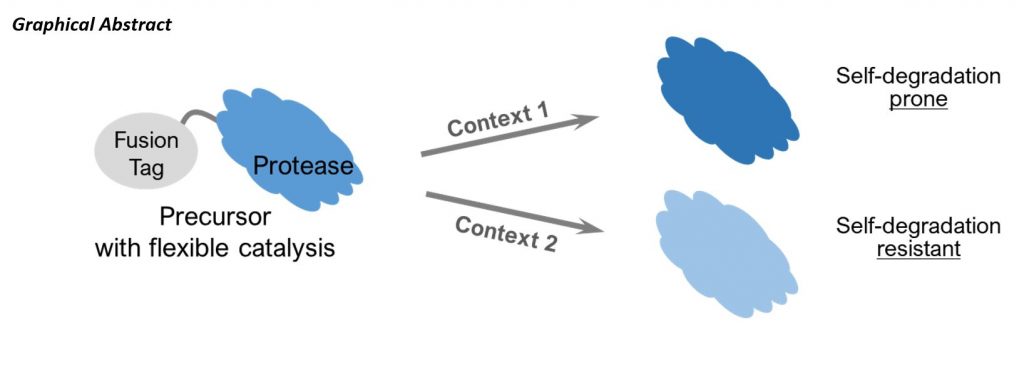
HIV protease inhibitor (PI) resistance compromises treatment efficacy and prognosis of combination antiretroviral therapy. The currently available PIs target the active site of mature protease (PR), however resistance-associated mutations (RAMs) identified in patients experiencing drug resistance map to various regions of protease. When introduced into E. coli for protein purification and in vitro characterization, many RAMs do not display drug resistance at the magnitudes observed in patients. We previously reported that precursor autoprocessing is context-dependent as its activity and outcomes can be modulated by sequences upstream of p6*-PR. Notably, the mature PRs released from select fusion precursors were self-degradation prone, resembling those purified from E. coli. In contrast, precursors with the 26aa maltose binding protein signal peptide (SigP) at the N-terminus released mature PRs resistant to self-degradation, resembling those found in viral particles. Therefore, SigP containing fusion precursors likely represent a more biologically relevant “context” and are expected to manifest the clinically observed drug resistance. To investigate this context, we engineered a panel of fusion precursor constructs containing either the wild-type or an indinavir resistance-associated double mutation (77I82T) in the PR region. Both wild type and 77I82T mutant precursors showed similar response to PI when released from constructs without SigP. However, the 77I82T mutant displayed a significant resistance profile when the N-terminal SigP was present in the precursors, which matches our viral infectivity analysis. These results support our context-dependent hypothesis and suggest that SigP-containing precursors better recapitulate PI response observed in a viral setting. This platform provides a unique insight for studying drug resistance.